Austin, TX, USA, Aug. 25, 2025 (GLOBE NEWSWIRE) — Custom Market Insights has published a new research report titled “Targeted Drug Delivery Market Size, Trends and Insights By Technology (Nanoparticle-Based Drug Delivery, Ligand-Based Targeting Systems, Polymeric Carriers, Antibody–Drug Conjugates (ADCs), Lipid-Based Delivery Systems), By Application (Oncology, Cardiovascular Diseases, Neurological Disorders, Infectious Diseases, Others), By End User (Hospitals & Clinics, Specialty & Cancer Treatment Centers, Research & Academic Institutes, Biopharmaceutical & Biotechnology Companies), and By Region – Global Industry Overview, Statistical Data, Competitive Analysis, Share, Outlook, and Forecast 2025 – 2034“ in its research database.
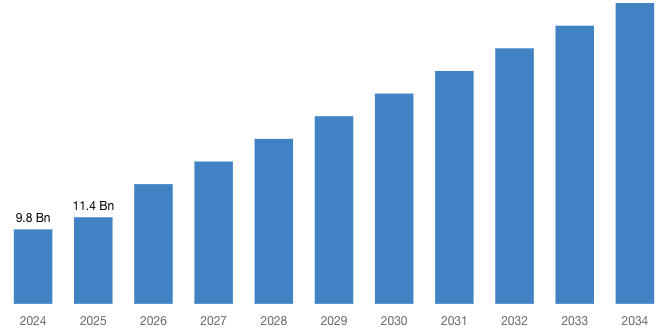
“According to the latest research study, the demand of the global Targeted Drug Delivery Market size & share was valued at approximately USD 9.8 Billion in 2024 and is expected to reach USD 11.4 Billion in 2025 and is expected to reach a value of around USD 39.6 Billion by 2034, at a compound annual growth rate (CAGR) of about 15.5% during the forecast period 2025 to 2034.”
Click Here to Access a Free Sample Report of the Global Targeted Drug Delivery Market @ https://www.custommarketinsights.com/request-for-free-sample/?reportid=72424
Overview
According to industry analysts at CMI, Targeted drug distribution opens up for the increasing occurrence of cancer, cardiovascular, or neurodegenerative disorders all around the world, creating demand for highly specific and effective treatment. Nanotechnology developments in ADCs (antibody-drug conjugates), ligand-based carriers, and lipid nanoparticles are improving their targeting ability, therapeutic efficacy, and safety profile.
The market is boosted by biologics, gene therapies, and mRNA drugs that need precise delivery to work effectively and avoid affecting other areas. Biomarker-driven patient selection and adoption are driving an increase in investment in personalized medicine. By having strategic collaborations globally between pharmaceutical companies, biotech firms, and research institutions, faster innovation is becoming a reality. Beneficial government policies, research grants, and regulatory incentives, such as orphan drug designations, are strengthening the R&D pipelines and thereby supporting global market growth.
Key Trends & Drivers
- Increasing Prevalence of Chronic Diseases: The growing prevalence of cancer, cardiovascular diseases, and neurological disorders is quite exorbitantly pushing the demand for targeted drug delivery systems. These platforms can include antibody–drug conjugates, nanocarriers, or ligand-based systems, which are capable of providing greater treatment accuracy with fewer side effects, thus enhancing patient compliance. Another typical example is that, as per WHO, cancer cases all over the globe will continue to rise; hence, treatment providers are increasingly relying on targeted therapies to improve patient outcomes while keeping treatment costs low. The increasing burden of these diseases would ensure the demand for site-specific, innovative delivery methods in the long term, ensuring that the market constantly grows in both developed and emerging economies during the forecast period.
- Technological Advancements in Delivery Systems: The continuous developments in nanotechnology, lipid nanoparticles, polymeric carriers, and ligand-based targeting have revolutionized precision in drug delivery. These technologies allow a drug to cross biological barriers, reach a specific tissue or cell, and achieve its payload release over time. Improvements in the biocompatibility, stability, and targeting efficiency have now set these technologies for applications beyond oncology to neurology, infectious diseases, and rare genetic disorders. The success of the COVID-19 vaccine using LNPs has showcased the ability for scale-up and global distribution, greatly accelerating R&D investment in this space. As emerging technologies start integrating AI with state-of-the-art material sciences, delivery platforms are becoming more efficient, adaptable, and patient-specific, contributing to faster adoption and higher clinical/commercial viability.
- The growth of biologics and gene therapies: including monoclonal antibodies, recombinant proteins, RNA-based drugs, and gene therapies, has resulted in a corresponding increase in the demand for targeted delivery systems. Many chemicals, being unstable or of a large size, require carriers to keep them bioavailable and acting in a targeted way. Gene therapy platforms such as CRISPR-Cas9 rely on the precise delivery mechanism of lipid nanoparticles or viral systems to be effective. Regulatory approvals for targeted biologics are growing in oncology and rare diseases, while continuous innovations are widening therapeutic coverage. This boom in biologics and the trend toward personalized medicine paints targeted delivery as the lifeblood of next-generation therapies.
Request a Customized Copy of the Targeted Drug Delivery Market Report @ https://www.custommarketinsights.com/request-for-customization/?reportid=72424
- R&D costs are high and a complex development process: Drug targeting systems often require complex design, sophisticated manufacture, and specialized tests concerning safety and efficacy, as well as meeting regulatory stipulations. Extensive preclinical and clinical studies often take several years during the developmental process. The risk-return profile of large investments, combined with the uncertainties of getting approvals, constitutes the major deterrent, especially for smaller biotechnology companies. Large pharmaceutical companies often fund these drug developments for many years, while innovators on the smaller side depend heavily on venture capital or strategic partnering. These economic and operational barriers slow entry into the market, reduce competition, and cluster innovation with a handful of large players that can afford the cost and risk.
- Rising Strategic Collaborations and Licensing Deals: Collaborations between pharmaceutical companies, biotech firms, academic institutions, and CROs are becoming increasingly important for advancing targeted drug delivery technologies. Such partnerships combine expertise with developers, shorten development timelines, and spread financial risk. Licensing deals, a classic example, allow companies to buy into patented technologies rather than develop them in-house. In the oncology field, these types of agreements are common as businesses require rapid innovation. Therefore, strategic partners can speed the development process to market worldwide, whereby one partner’s regulatory, manufacturing, and distribution channels can be tapped into to aid in market expansion and therapeutic acceptance.
- Regulatory Landscape and Market Access: Regulatory bodies like the FDA and EMA hold a decisive place in shaping the targeted drug delivery market. Heavy approval processes ensure safety but delay time to market. Incentive schemes, such as orphan drug designation, accelerated approval, and compassionate use, facilitate faster market entry for therapies that are in high demand. In emerging markets, regulatory landscapes evolve to widen access, yet the infrastructure challenges can dwarf the adoption. Successfully weaving through the regulatory pathways with strong clinical evidence would be paramount for a therapy to establish itself in the market, especially when dealing with pricey treatments, where payers’ acceptance drives commercial success.
Report Scope
| Feature of the Report | Details |
| Market Size in 2025 | USD 11.4 Billion |
| Projected Market Size in 2034 | USD 39.6 Billion |
| Market Size in 2024 | USD 9.8 Billion |
| CAGR Growth Rate | 15.5% CAGR |
| Base Year | 2024 |
| Forecast Period | 2025-2034 |
| Key Segment | By Technology, Application, End User and Region |
| Report Coverage | Revenue Estimation and Forecast, Company Profile, Competitive Landscape, Growth Factors and Recent Trends |
| Regional Scope | North America, Europe, Asia Pacific, Middle East & Africa, and South & Central America |
| Buying Options |